Although cutaneous melanoma and uveal melanoma both arise from melanocytes, they differ fundamentally in genetics, biology, and response to therapies. Understanding these differences is central to developing more effective, targeted treatments for uveal melanoma.1,2
Uveal melanoma has a low tumor mutational burden, unlike cutaneous melanoma, likely contributing to its poor immunotherapy responses1,3,4
Low mutational burden in uveal melanoma results in a lack of neoantigens, rendering the tumor less perceptible to the immune system and contributing to an "immune-cold" microenvironment. Furthermore, the liver, which is the primary site of metastasis in 90% of cases, fosters an immunosuppressive microenvironment that further hampers immune recognition.1,3-6
As such, therapies that have revolutionized metastatic cutaneous melanoma treatment, such as checkpoint inhibitors, have limited improvement in survival and response rates in metastatic uveal melanoma.1,2,5,7
GNAQ/11 mutations are the primary oncogenic drivers in uveal melanoma5,8
Unlike cutaneous melanoma, in which BRAF and NRAS mutations are common, activating mutations in the genes coding for GNAQ or GNA11 occur in ~90% of uveal melanoma patients. Other less common driver mutations include CYSLTR2 or PLCβ4.9,10
Mutations in GNAQ or GNA11 lock them in a constitutively active state, triggering multiple downstream signaling cascades that are critical for uveal melanoma development and progression.1,11,12
These mutations impact the GPCR signaling pathway, resulting in PKC pathway overactivation, and are responsible for the oncogenesis of nearly all cases of uveal melanoma.5,10,13

Molecular pathways in uveal melanoma development and progression
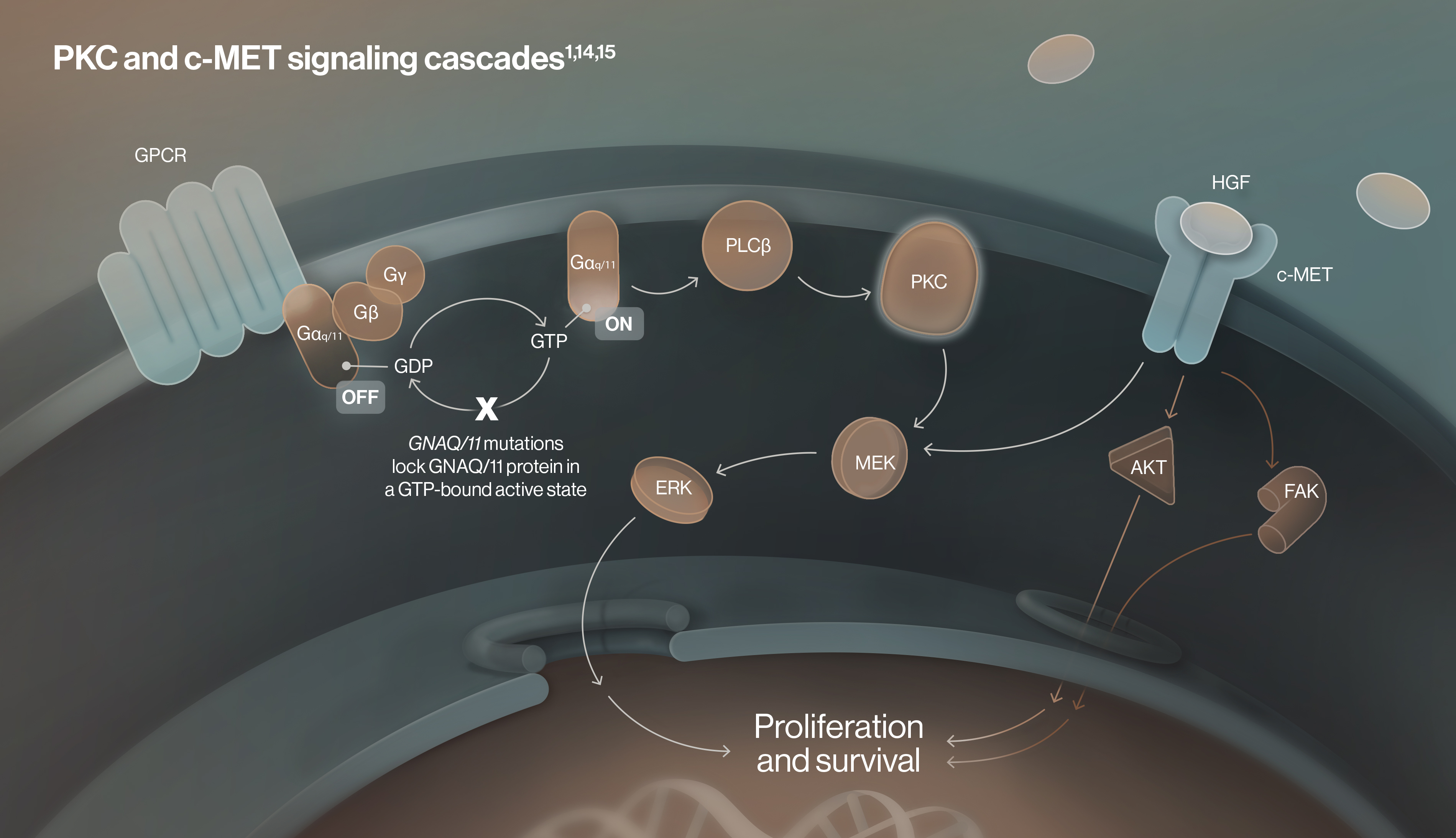
Overactive PKC signaling drives uveal melanoma cell proliferation and survival13
Mutant GNAQ or GNA11 stimulates PLCβ to activate PKC which, in turn, stimulates the MAPK pathway (MEK/ERK) driving uveal melanoma cell proliferation and survival.1,11,15,16
Loss of BAP1 function, commonly observed in uveal melanoma, affects DNA integrity regulation, cell division, and melanoma cell survival, processes whose disruption plays a role in oncogenic transformation.1
c-MET overexpression and pathway overactivation are associated with uveal melanoma tumor metastasis17,18
In metastatic uveal melanoma, elevated HGF and overexpression of its receptor c-MET in the metastatic tumor microenvironment further activate signaling pathways to sustain metastatic growth.17,18
HGF/c-MET signaling may mediate resistance to therapies and sustain tumor survival.17,19
Collectively, GNAQ/GNA11-driven PKC activation and HGF/c-MET-mediated signaling represent critical pathways to consider in the management and treatment of metastatic uveal melanoma.17,18
Meet
[Dr. Smith]
[Dr. Smith] provides insights into uveal melanoma, exploring its unique biology, clinical
challenges, and current approaches to diagnosis and management.

Grounded in an understanding of metastatic uveal melanoma biology, IDEAYA Biosciences leverages scientific insight to drive innovation and advance care.
See how IDEAYA Biosciences is advancing innovation in metastatic uveal melanoma

BAP1, BRCA1-associated protein 1; BRAF, B-Raf proto-oncogene; c-MET, mesenchymal epithelial transition factor; CYSLTR2, cysteinyl leukotriene receptor 2; DNA, deoxyribonucleic acid; ERK, extracellular signal-regulated kinase; GNA11, G protein subunit alpha-11; GNAQ, G protein subunit alpha-q; GPCR, G protein-coupled receptor; HGF, hepatocyte growth factor; MAPK, mitogen-activated protein kinase; MEK, mitogen-activated protein kinase kinase; NRAS, neuroblastoma rat sarcoma (RAS) viral oncogene homolog; PKC, protein kinase C; PLCβ4, phospholipase C beta 4.
1. Pandiani C, Béranger GE, Leclerc J, Ballotti R, Bertolotto C. Focus on cutaneous and uveal melanoma specificities. Genes Dev. 2017;31(8):724-743. doi:10.1101/gad.296962.117
2. Koch EAT, Heppt MV, Berking C. The current state of systemic therapy of metastatic uveal melanoma. Am J Clin Dermatol. 2024;25(5):691-700. doi:10.1007/s40257-024-00872-1
3. Song J, Mou P, Song GG, Chen L, Chen YQ, Wei RL. Advances in immunotherapy for uveal melanoma: enhancing efficacy and overcoming resistance. Front Cell Dev Biol. 2025;13:1619150. doi:10.3389/fcell.2025.1619150
4. Wessely A, Steeb T, Erdmann M, et al. The role of immune checkpoint blockade in uveal melanoma. Int J Mol Sci. 2020;21(3):879. doi:10.3390/ijms21030879
5. Park JJ, Diefenbach RJ, Joshua AM, Kefford RF, Carlino MS, Rizos H. Oncogenic signaling in uveal melanoma. Pigment Cell Melanoma Res. 2018;31(6):661-672. doi:10.1111/pcmr.12708
6. Diener-West M, Reynolds SM, Agugliaro DJ, et al. Development of metastatic disease after enrollment in the COMS trials for treatment of choroidal melanoma: collaborative Ocular Melanoma Study Group Report No. 26. Arch Ophthalmol. 2005;123(12):1639-1643. doi:10.1001/archopht.123.12.1639
7. Mallone F, Sacchetti M, Lambiase A, Moramarco A. Molecular insights and emerging strategies for treatment of metastatic uveal melanoma. Cancers (Basel). 2020;12(10):2671. doi:10.3390/cancers12102761
8. Carvajal RD, Sacco JJ, Jager MJ, et al. Advances in the clinical management of uveal melanoma. Nat Rev Clin Oncol. 2023;20(2):99-115. doi:10.1038/s41571-022-00714-1
9. Silva-Rodríguez P, Fernández-Díaz D, Bande M, Pardo M, Loidi L, Blanco-Teijeiro MJ. GNAQ and GNA11 genes: a comprehensive review on oncogenesis, prognosis and therapeutic opportunities in uveal melanoma. Cancers (Basel). 2022;14(13):3066. doi:10.3390/cancers14133066
10. Robertson AG, Shih J, Yau C, et al. Integrative analysis identifies four molecular and clinical subsets in uveal melanoma. Cancer Cell. 2017;32(2):204-220.e15. doi:10.1016/j.ccell.2017.07.003
11. Carvajal RD, Schwartz GK, Tezel T, Marr B, Francis JH, Nathan PD. Metastatic disease from uveal melanoma: treatment options and future prospects. Br J Ophthalmol. 2017;101(1):38-44. doi:10.1136/bjophthalmol-2016-309034
12. Romano E, Schwartz GK, Chapman PB, Wolchock JD, Carvajal RD. Treatment implications of the emerging molecular classification system for melanoma. Lancet Oncol. 2011;12(9):913-922. doi:10.1016/S1470-2045(10)70274-6
13. Park JJ, Abou Hamad S, Stewart A, Carlino MS, Lim SY, Rizos H. PKC-independent PI3K signaling diminishes PKC inhibitor sensitivity in uveal melanoma. Oncogenesis. 2024;13(1):9. doi:10.1038/s41389-024-00511-8
14. Wespiser M, Neidhardt E, Negrier S. Uveal melanoma: in the era of new treatments. Cancer Treat Rev. 2023;119:102599. doi:10.1016/j.ctrv.2023.102599
15. Chen X, Wu Q, Depeille P, et al. RasGRP3 mediates MAPK pathway activation in GNAQ mutant uveal melanoma. Cancer Cell. 2017;31(5):685-696.e6. doi:10.1016/j.ccell.2017.04.002
16. Wu X, Li J, Zhu M, Fletcher JA, Hodi FS. Protein kinase C inhibitor AEB071 targets ocular melanoma harboring GNAQ mutations via effects on the PKC/ERK1/2 and PKC/NF-kB Pathways. Mol Cancer Ther. 2012;11(9):1905-1914. doi:10.1158/1535-7163.MCT-12-0121
17. Tanaka R, Terai M, Londin E, Sato T. The role of HGF/MET signaling in metastatic uveal melanoma. Cancers (Basel). 2021;13(21):5457. doi:10.3390/cancers13215457
18. Surriga O, Rajasekhar VK, Ambrosini G, Dogan Y, Huang R, Schwartz GK. Crizotinib, a c-Met inhibitor, prevents metastasis in a metastatic uveal melanoma model. Mol Cancer Ther. 2013;12(12):2817-2826. doi:10.1158/1535-7163.MCT-13-0499
19. Khan S, Lutzky J, Shoushtari AN, et al. Adjuvant crizotinib in high-risk uveal melanoma following definitive therapy. Front Oncol. 2022;12:976837. doi:10.3389/fonc.2022.976837
